Uranium atomic structure12/22/2023  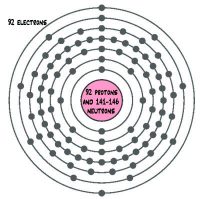
It could also help more effectively manage nuclear waste. This is an important step toward advancing the design of next-generation nuclear materials. The results of this study help scientists understand and predict the chemical and physical behavior of transuranic elements-elements beyond uranium in the periodic table. This shell is also the region of mismatch between scientists’ observations and theories of how electrons are organized around atomic nuclei. These covalent bonds correlated with the 5 f shell of the quantum-mechanical model of the atom. The researchers probed the anion’s electronic structure and found that while the electrons in the plutonium-chlorine bonds were mostly not shared (ionic), they also featured important contributions from covalent bonds where the electrons are shared. Researchers synthesized five different hybrid materials containing a particular compound subunit of plutonium and chlorine: the 2- anion. Also, these elements, which are in a sequence of related elements called the actinide series, have electrons that are organized in ways that don’t match theoretical predictions. These elements are challenging to study due to their complex chemistry and radioactivity. The electronic configuration of Uranium will be 1s2 2s2 2p6 3s2 3p6 3d10 4s2 4p6 4d10 5s2 5p6 4f14 5d10 6s2 6p6 5f3 6d1 7s2.Understanding the arrangement of electrons in compounds containing actinide elements, such as uranium and plutonium, can help advance the design of next-generation nuclear materials. How do you write the electron configuration for Uranium? What is the electronic configuration of Uranium 92? What is the boiling Point of Uranium in Kelvin?īoiling Point of Uranium in Kelvin is 4200 K. Melting Point of Uranium in Kelvin is 1408 K. What is the melting Point of Uranium in Kelvin? What is the boiling Point of Uranium?īoiling Point of Uranium is 4200 K. Uranium has 92 electrons out of which 6 valence electrons are present in the 5f3 6d1 7s2 outer orbitals of atom. How many valence electrons does a Uranium atom have? 
It is located in group null and period 7 in the modern periodic table. Uranium is the 92 element on the periodic table. Uranium is a chemical element with the symbol U and atomic number 92. What is the position of Uranium in the Periodic Table? 
Uranium is a chemical element with symbol U and atomic number 92. To form abbreviated notation of electronic configuration, the completely filled subshells are replaced by the noble gas of the preceding period in square brackets. The abbreviated electronic configuration of Uranium is 5f3 6d1 7s2. What is the abbreviated electronic configuration of Uranium? What is the electronic configuration of Uranium? Uranium Thermal Properties - Enthalpies and thermodynamics Optical Properties of Uranium Refractive IndexĪcoustic Properties of Uranium Speed of Sound 
Uranium Magnetic Properties Magnetic Type Uranium Heat and Conduction Properties Thermal Conductivity Refer to table below for the Electrical properties ofUranium Electrical Conductivity Hardness of Uranium - Tests to Measure of Hardness of Element Mohs Hardness Refer to below table for Uranium Physical Properties Densityġ9.05 g/cm3(when liquid at m.p density is $17.3 g/cm3) 
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
